Covid-19 Antigen Rapid Test Device
The Rapid Response covid-19 antigen rapid test device is an in vitro immunochromatographic assay for the direct and qualitative detection of SARS-CoV-2 viral nucleoprotein antigens from nasal and nasopharyngeal secretions from individuals suspected of COVID-19 within 6 days of symptom onset.
This test is authorized for use at the Point of Care i.e., in a patient care setting.
An antigen is generally detectable in nasopharyngeal and nasal secretions during the acute phase of infection.
Pursuant to section 5 of the Interim Order Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19, made by the Minister of Health on March 18, 2020, the Rapid Response COVID-19 Antigen Rapid Test Device is now authorized for sale or importation in Canada.
*Upload test results to your database using RapidReader App.
POINTS TO REMEMBER
- Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
- Positive results do not rule out bacterial infection or co-infection with other viruses
- Laboratories are required to report all positive results to the appropriate public health authorities
- Negative results should be treated as presumptive, and do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions
- Negative results should be considered in the context of a patient’s recent exposures, history, and the presence of clinical signs and symptoms consistent with COVID-19, and confirmed with a molecular assay, if necessary, for patient management

Product Information
Product Information
Shipping & Returns
Shipping & Returns
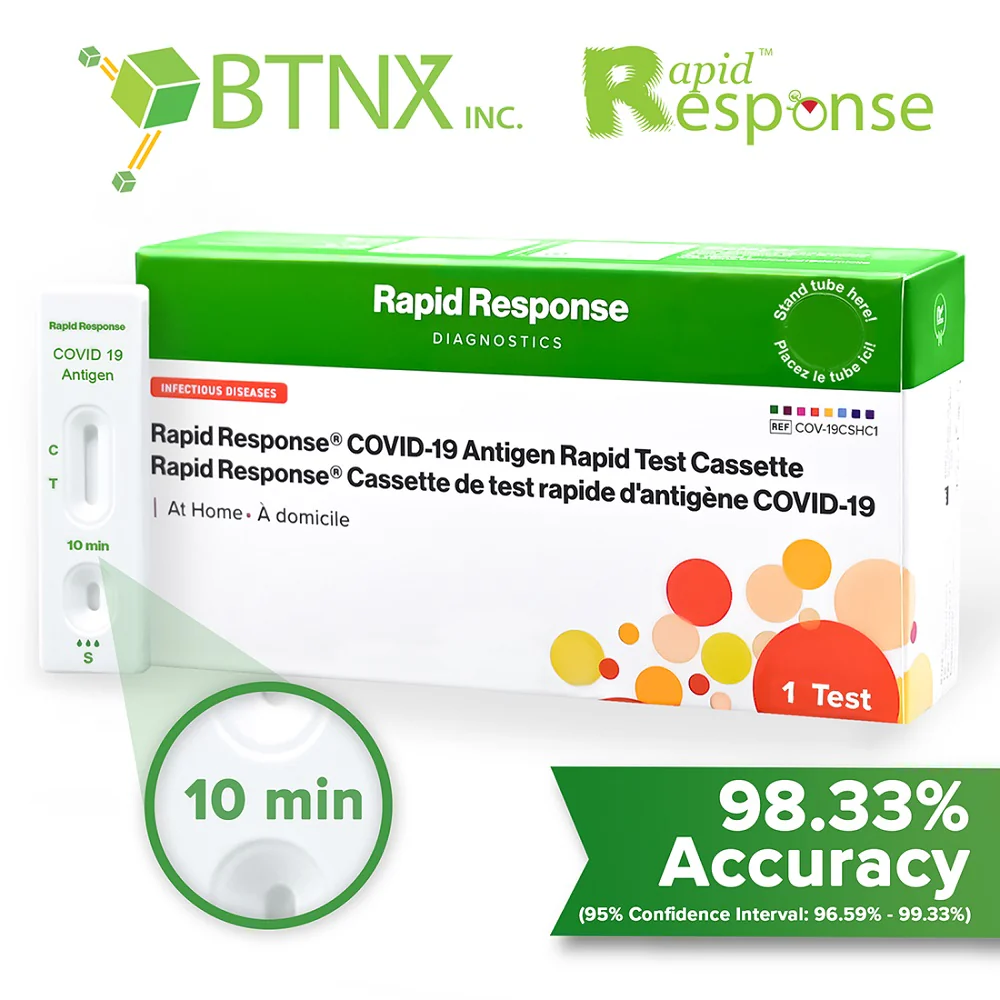
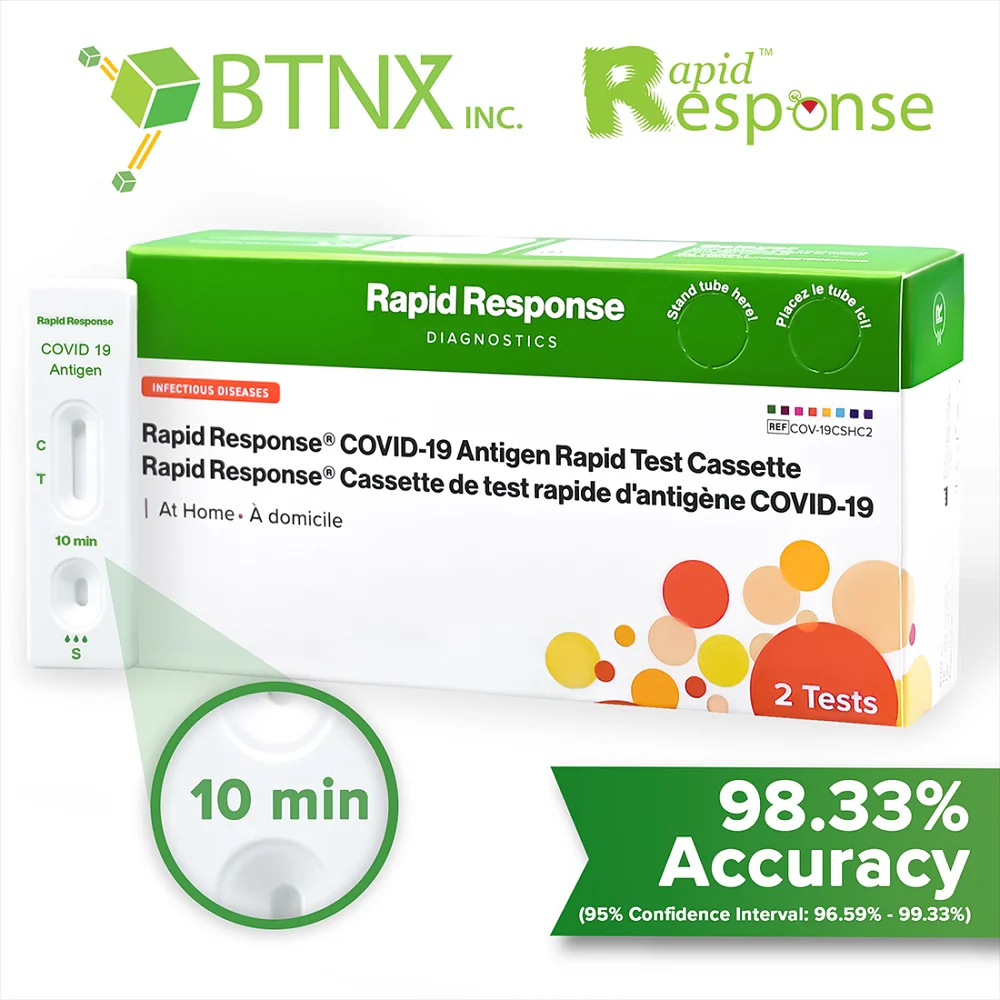






Covid-19 Antigen Rapid Test Device
Covid-19 Antigen Rapid Test Device
The Rapid Response covid-19 antigen rapid test device is an in vitro immunochromatographic assay for the direct and qualitative detection of SARS-CoV-2 viral nucleoprotein antigens from nasal and nasopharyngeal secretions from individuals suspected of COVID-19 within 6 days of symptom onset.
This test is authorized for use at the Point of Care i.e., in a patient care setting.
An antigen is generally detectable in nasopharyngeal and nasal secretions during the acute phase of infection.
Pursuant to section 5 of the Interim Order Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19, made by the Minister of Health on March 18, 2020, the Rapid Response COVID-19 Antigen Rapid Test Device is now authorized for sale or importation in Canada.
*Upload test results to your database using RapidReader App.
POINTS TO REMEMBER
- Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
- Positive results do not rule out bacterial infection or co-infection with other viruses
- Laboratories are required to report all positive results to the appropriate public health authorities
- Negative results should be treated as presumptive, and do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions
- Negative results should be considered in the context of a patient’s recent exposures, history, and the presence of clinical signs and symptoms consistent with COVID-19, and confirmed with a molecular assay, if necessary, for patient management

Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
The Rapid Response covid-19 antigen rapid test device is an in vitro immunochromatographic assay for the direct and qualitative detection of SARS-CoV-2 viral nucleoprotein antigens from nasal and nasopharyngeal secretions from individuals suspected of COVID-19 within 6 days of symptom onset.
This test is authorized for use at the Point of Care i.e., in a patient care setting.
An antigen is generally detectable in nasopharyngeal and nasal secretions during the acute phase of infection.
Pursuant to section 5 of the Interim Order Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19, made by the Minister of Health on March 18, 2020, the Rapid Response COVID-19 Antigen Rapid Test Device is now authorized for sale or importation in Canada.
*Upload test results to your database using RapidReader App.
POINTS TO REMEMBER
- Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
- Positive results do not rule out bacterial infection or co-infection with other viruses
- Laboratories are required to report all positive results to the appropriate public health authorities
- Negative results should be treated as presumptive, and do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions
- Negative results should be considered in the context of a patient’s recent exposures, history, and the presence of clinical signs and symptoms consistent with COVID-19, and confirmed with a molecular assay, if necessary, for patient management