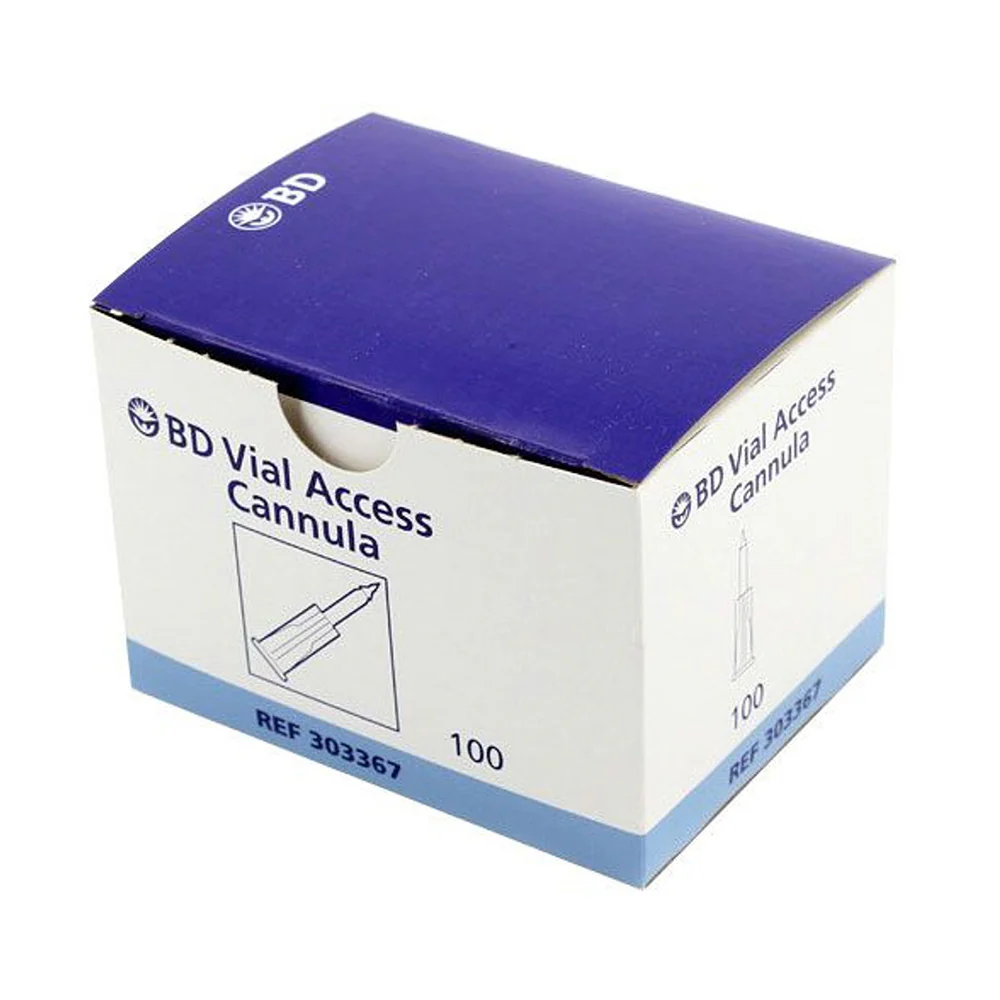
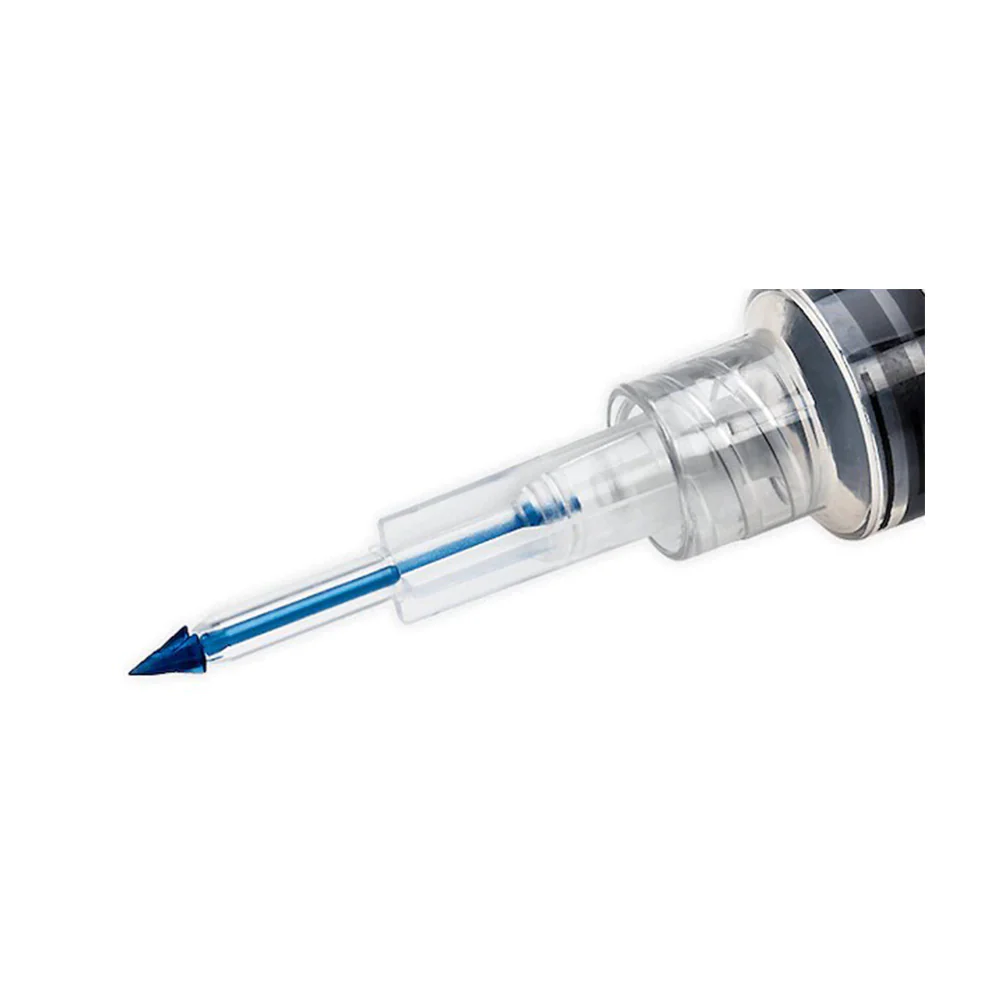
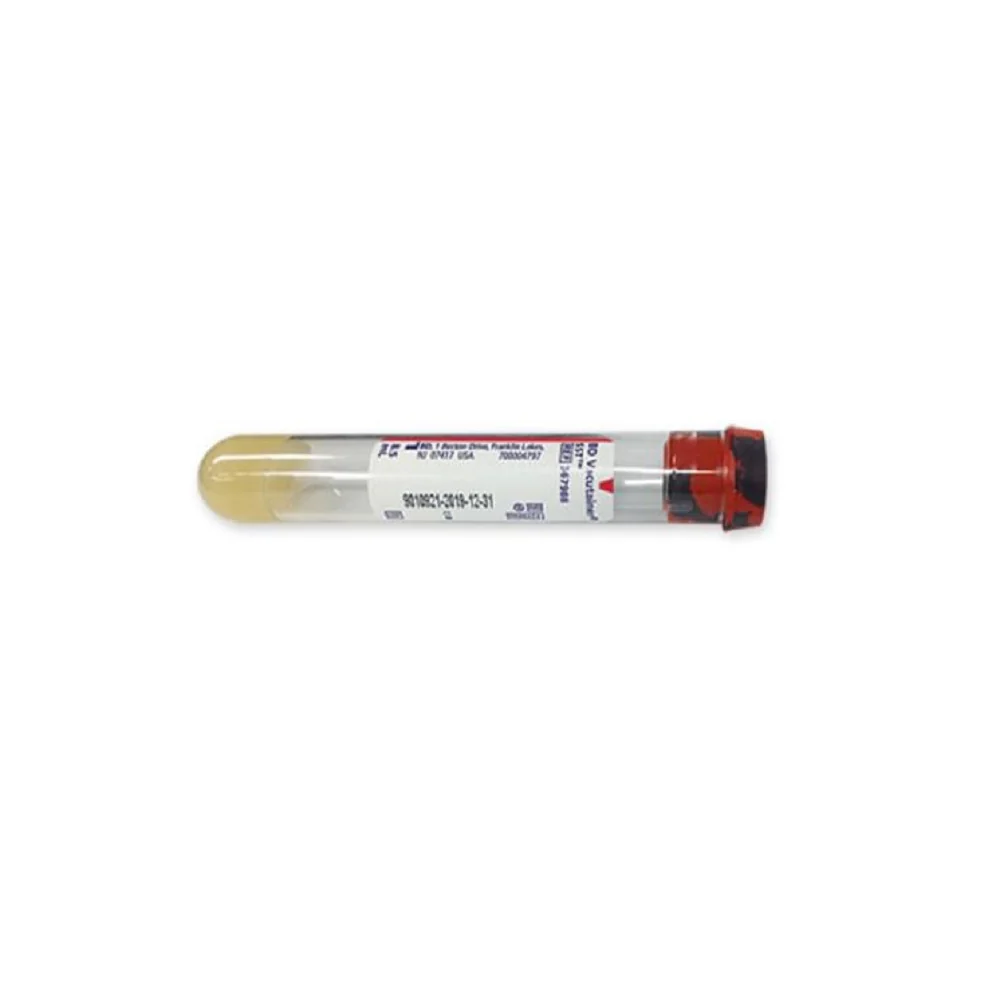
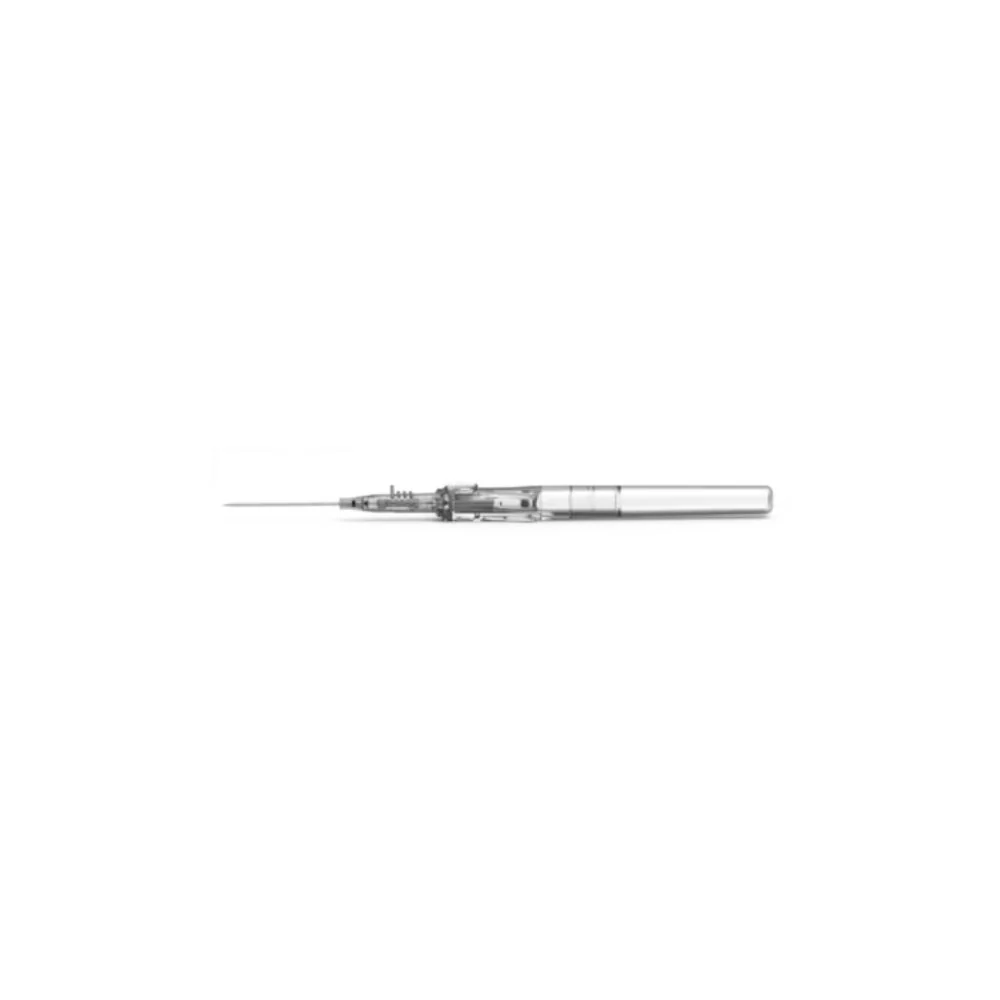
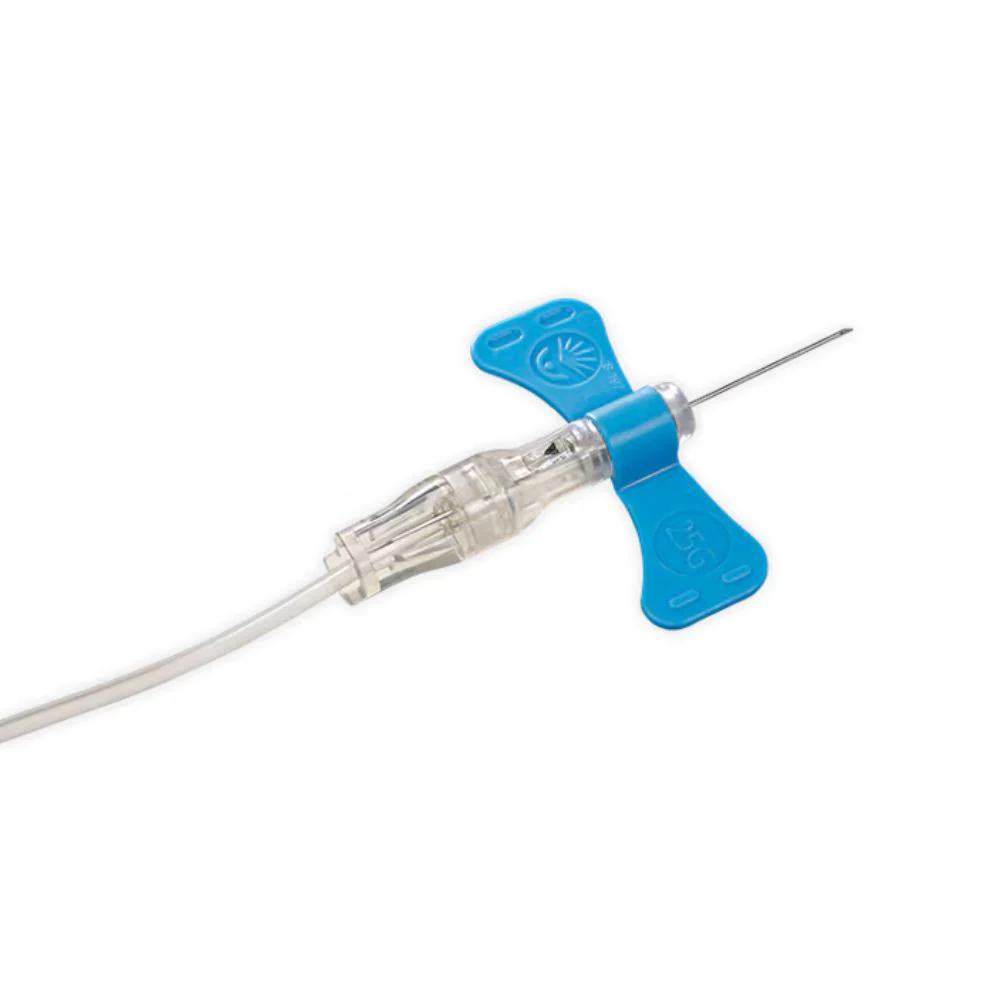
BD Vacutainer® Blood Transfer Device - Case of 198
The BD Vacutainer® blood transfer device eliminates the hazardous practice of needle-based specimen transfer, addressing both occupational safety concerns and regulatory compliance requirements. This single-use mechanism facilitates safe conveyance of venous blood from syringes to collection tubes and culture bottles without compromising sample quality or analytical validity. By removing direct needle handling during the transfer process, the device substantially reduces percutaneous injury risk to laboratory and clinical personnel. Supplied in a case of 198 units, this product supports consistent adherence to safety protocols across high-volume specimen processing operations.
Product Information
Product Information
Shipping & Returns
Shipping & Returns

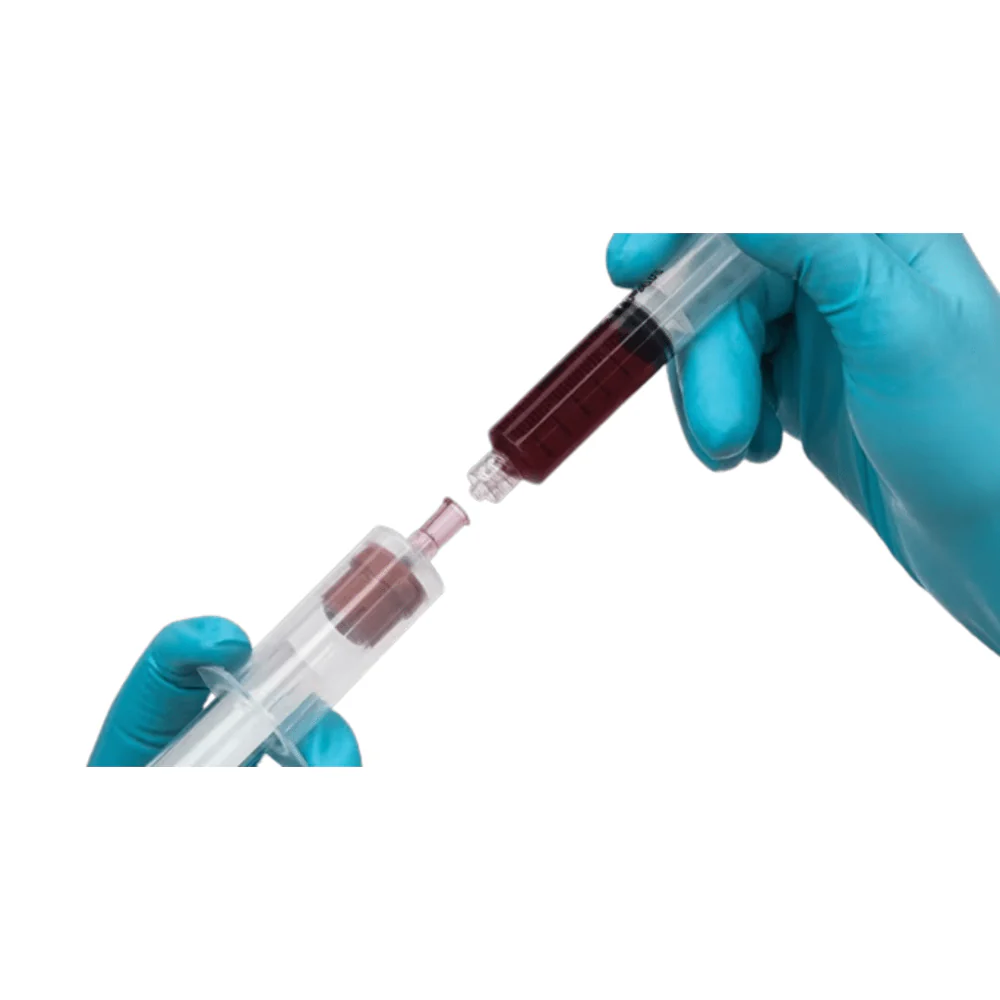
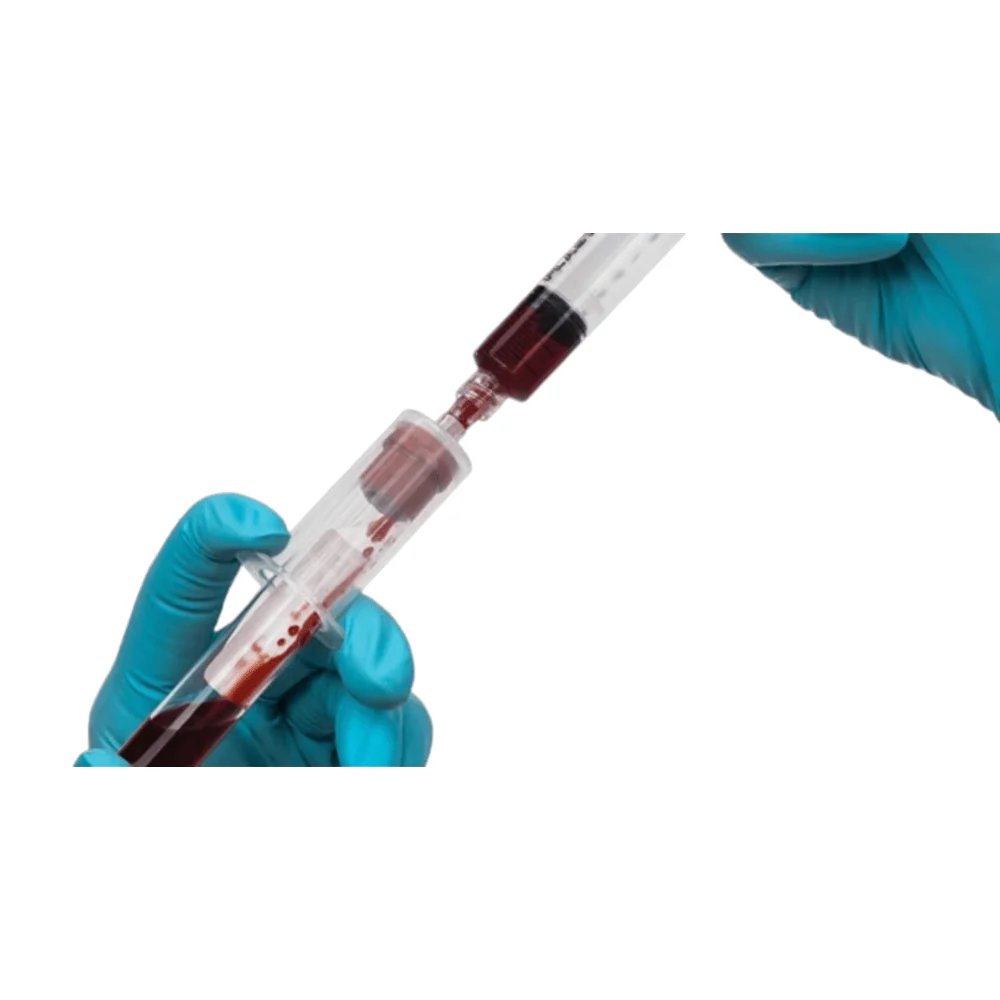
BD Vacutainer® Blood Transfer Device - Case of 198
BD Vacutainer® Blood Transfer Device - Case of 198
The BD Vacutainer® blood transfer device eliminates the hazardous practice of needle-based specimen transfer, addressing both occupational safety concerns and regulatory compliance requirements. This single-use mechanism facilitates safe conveyance of venous blood from syringes to collection tubes and culture bottles without compromising sample quality or analytical validity. By removing direct needle handling during the transfer process, the device substantially reduces percutaneous injury risk to laboratory and clinical personnel. Supplied in a case of 198 units, this product supports consistent adherence to safety protocols across high-volume specimen processing operations.
Original: $292.56
-65%$292.56
$102.40Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
The BD Vacutainer® blood transfer device eliminates the hazardous practice of needle-based specimen transfer, addressing both occupational safety concerns and regulatory compliance requirements. This single-use mechanism facilitates safe conveyance of venous blood from syringes to collection tubes and culture bottles without compromising sample quality or analytical validity. By removing direct needle handling during the transfer process, the device substantially reduces percutaneous injury risk to laboratory and clinical personnel. Supplied in a case of 198 units, this product supports consistent adherence to safety protocols across high-volume specimen processing operations.